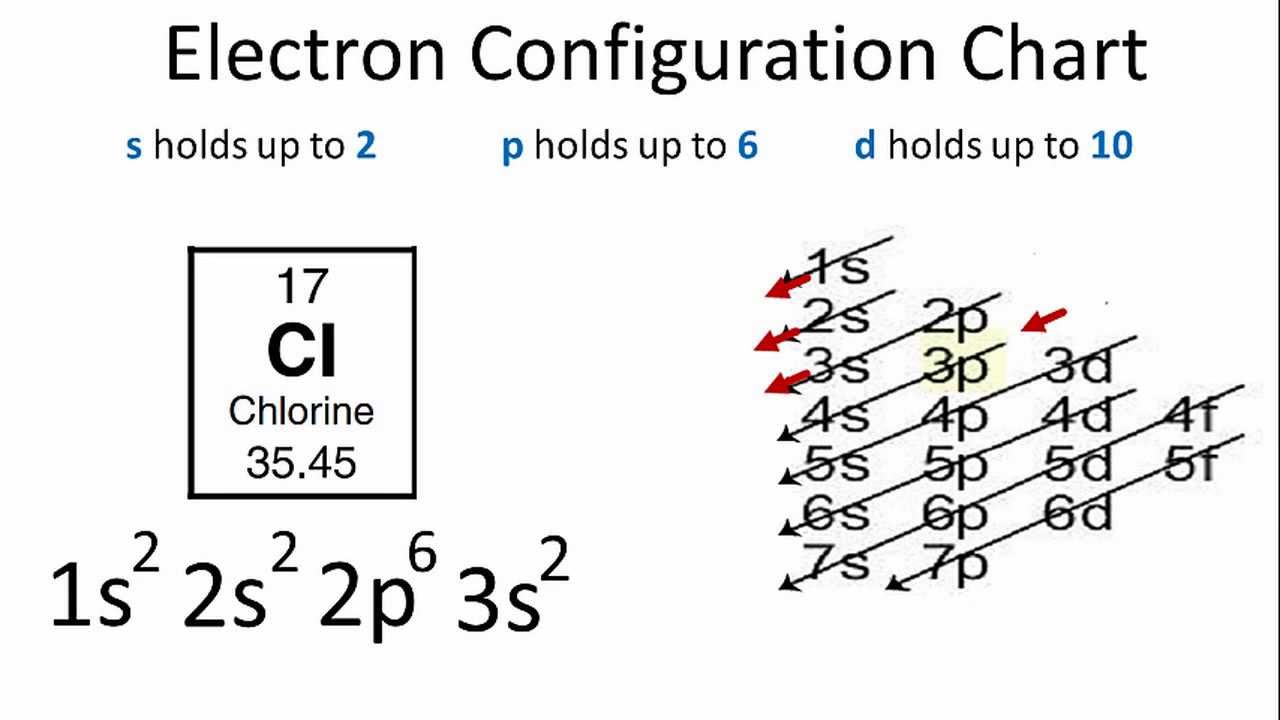
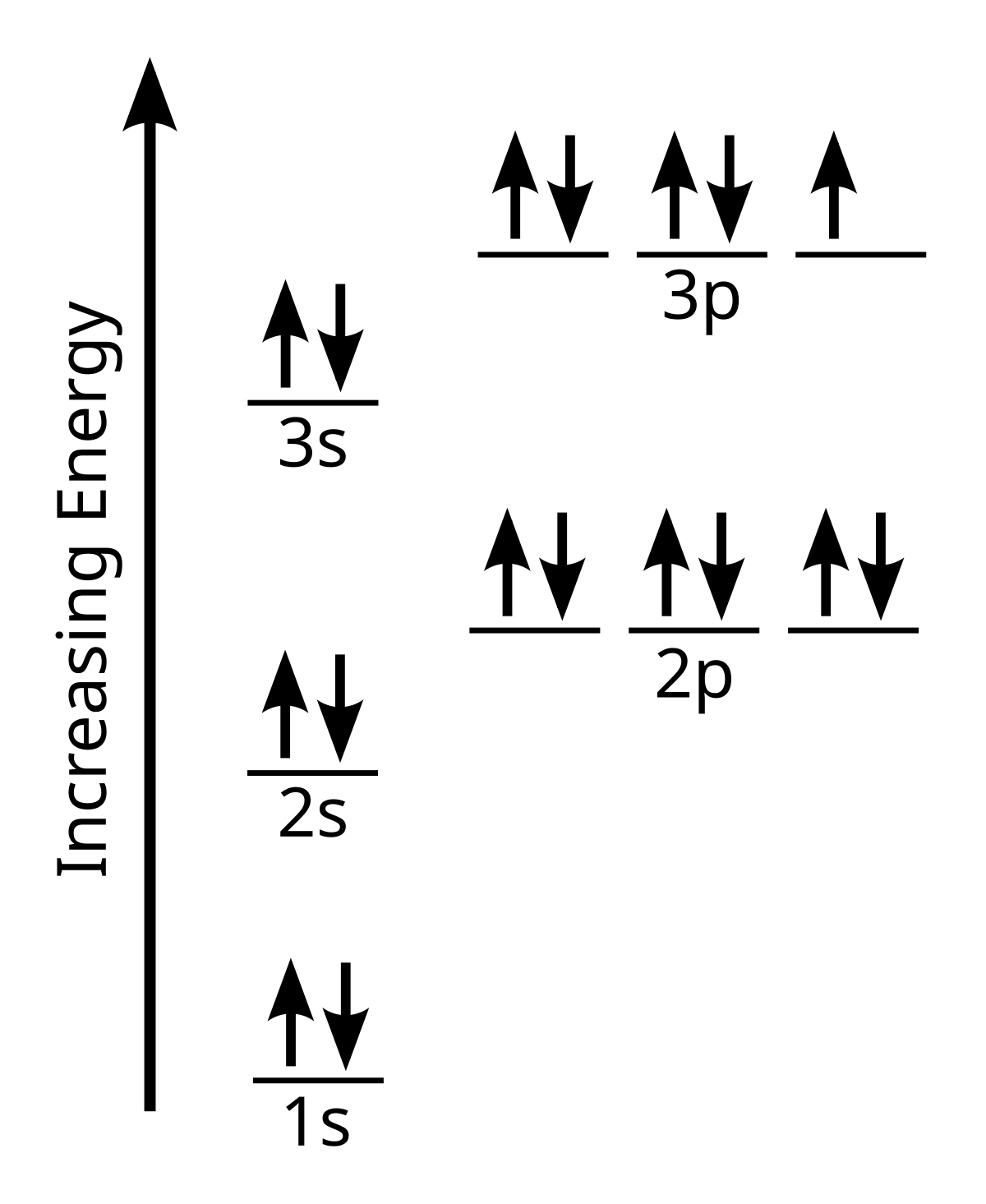
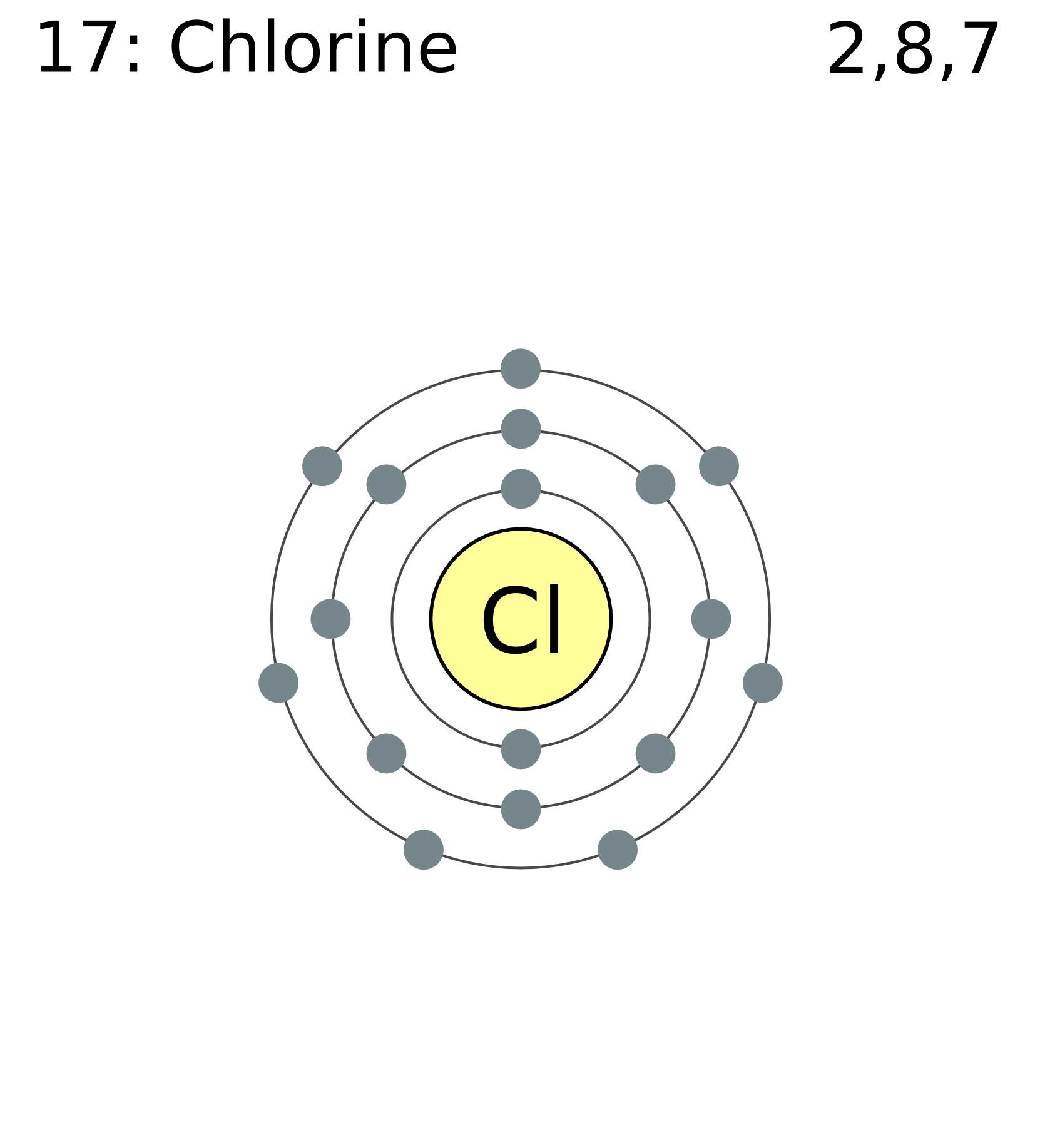
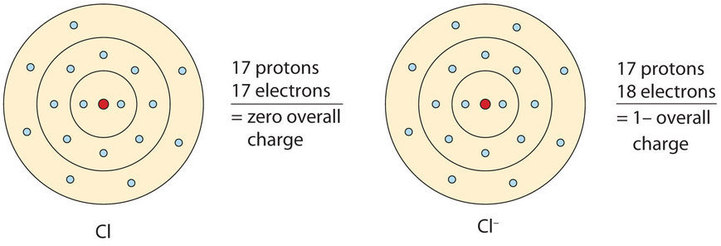
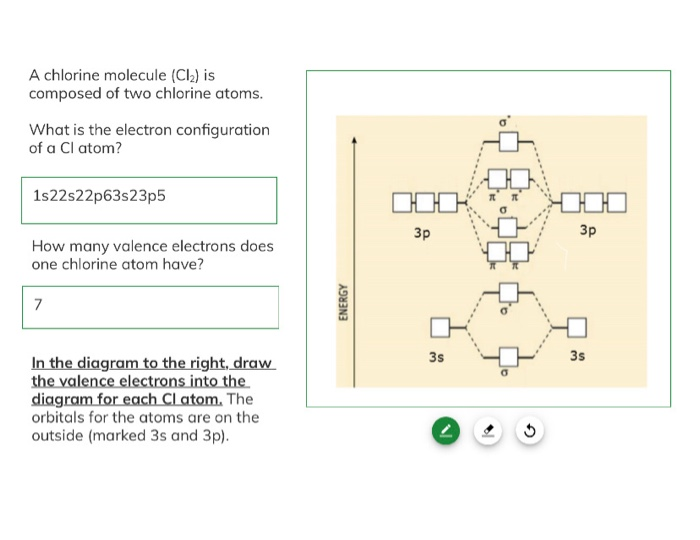
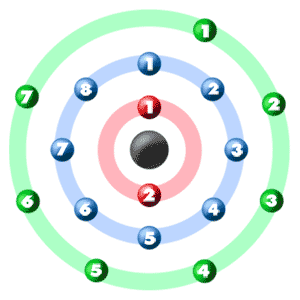
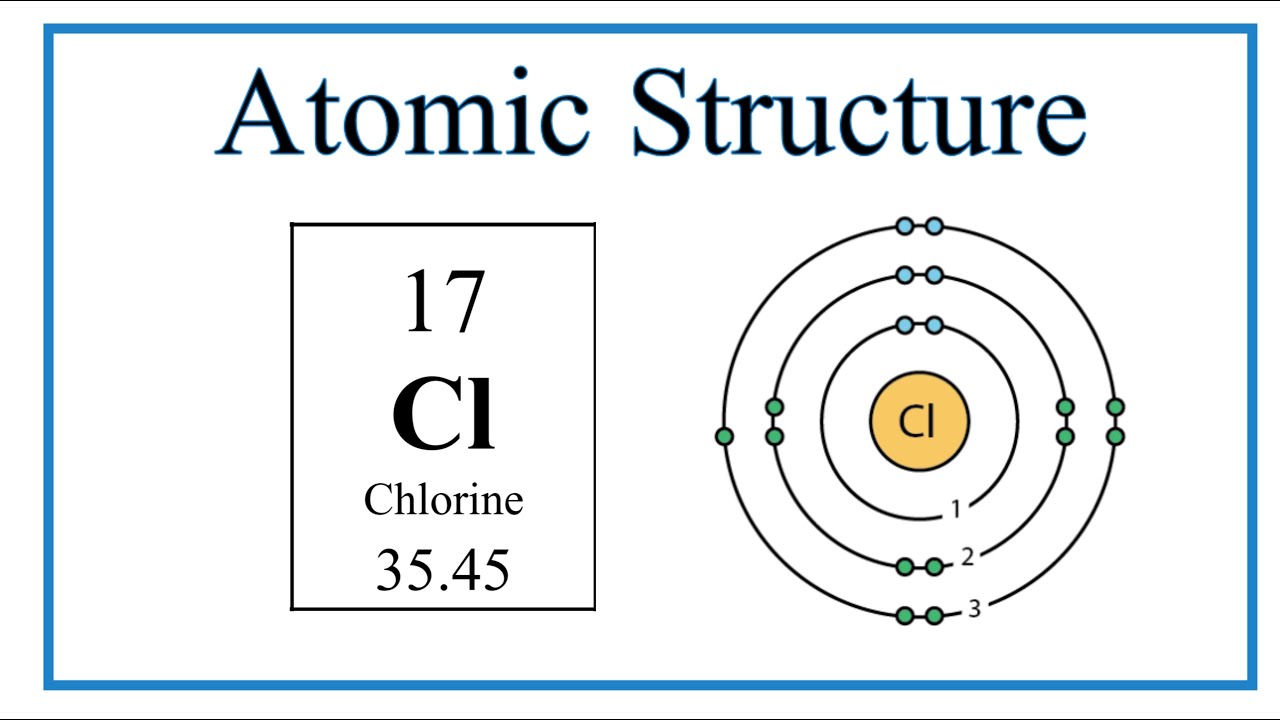
I just read that Chlorine(17) is missing 1 valence electron, but the third shell can hold up to 18 electrons, so isn't it missing 11? - Quora
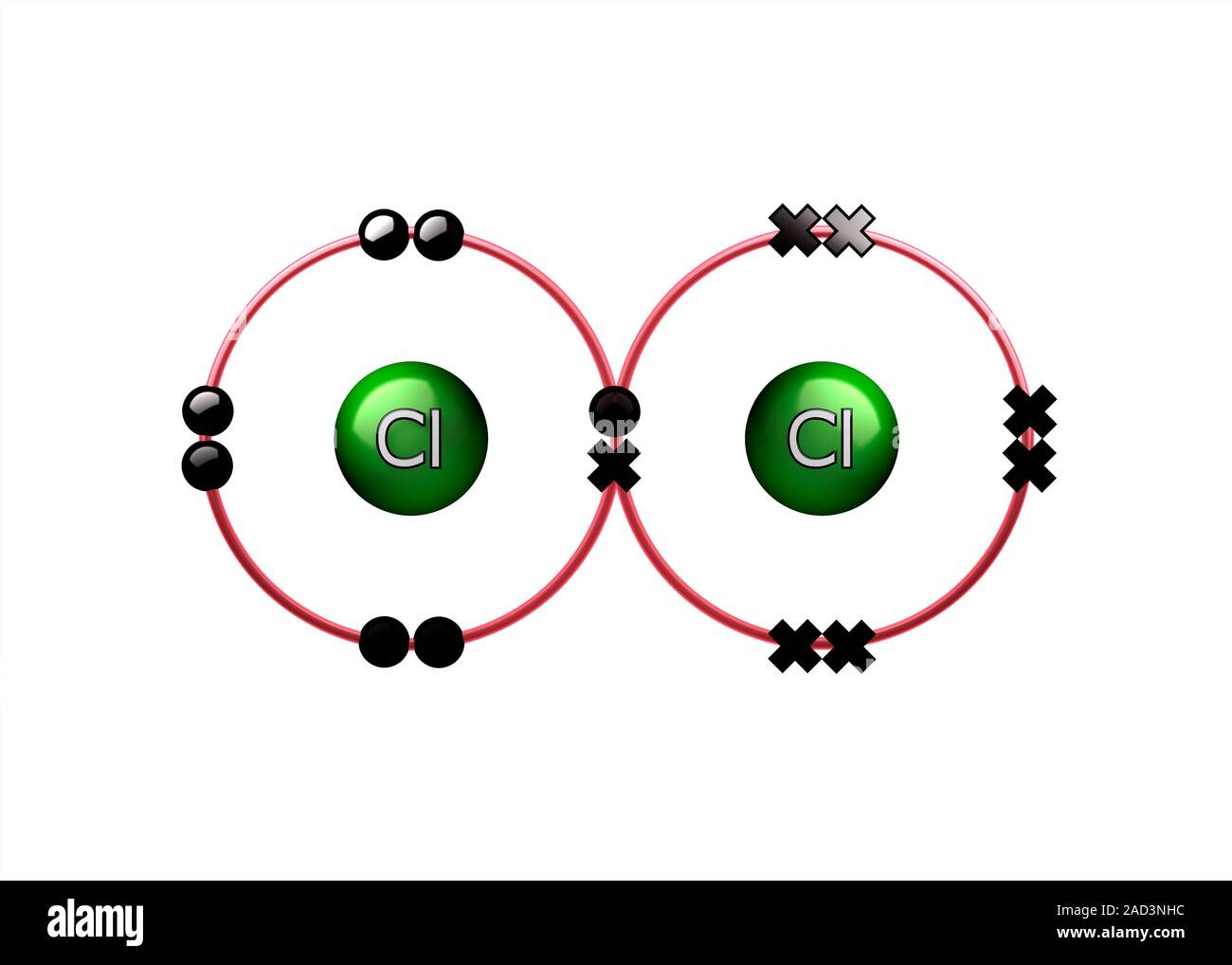
Bond formation in chlorine molecule. Illustration of the sharing of electrons (dots and crosses) between two chlorine atoms (Cl) to form a molecule of Stock Photo - Alamy